| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://www.theijcp.org |
Case Report
Volume 13, Number 2, June 2024, pages 48-53
Perioperative Care of a Child With Allan-Herndon-Dudley Syndrome
Wajahat Nazir Khana, Renee Henga, b, Joseph D. Tobiasa, b, c
aDepartment of Anesthesiology and Pain Medicine, Nationwide Children’s Hospital, Columbus, OH, USA
bDepartment of Anesthesiology and Pain Medicine, The Ohio State University College of Medicine, Columbus, OH, USA
cCorresponding Author: Joseph D. Tobias, Department of Anesthesiology and Pain Medicine, Nationwide Children’s Hospital, Columbus, OH 43205, USA
Manuscript submitted April 3, 2024, accepted June 7, 2024, published online June 23, 2024
Short title: Anesthesia and Allan-Herndon-Dudley syndrome
doi: https://doi.org/10.14740/ijcp535
| Abstract | ▴Top |
Allan-Herndon-Dudley syndrome (AHDS) is a rare X-linked disorder resulting from mutation of the SLC16A2 gene leading to a deficiency of a the monocarboxylate transporter 8 (MCT8) protein, which facilitates the passage of triiodothyronine (T3) into the central nervous system (CNS). Central lack of T3 leads to increased production of thyroid-stimulating hormone (TSH) with peripheral overproduction of thyroid hormones and systemic signs and symptoms of hyperthyroidism, as well as severe CNS effects including neurodevelopment delay, intellectual disability, hypotonia, and failure to thrive. We present a 6-year-old child with AHDS who required multiple anesthetics for various procedures, including right open adductor tenotomy, right proximal femoral varus osteotomy, and right acetabular osteotomy. The basic cellular defect of AHDS is described, end-organ effects outlined, and impact on perioperative care reviewed.
Keywords: Thyroid hormones; Thyroxine; Triiodothyronine; Hypothyroidism; Hypotonia
| Introduction | ▴Top |
Allan-Herndon-Dudley syndrome (AHDS), also known as MCT8 (monocarboxylate transporter 8) deficiency, is a rare genetic disorder resulting from a defect in the transport of thyroid hormone into the central nervous system (CNS) due to a lack of thyroid hormone transporter protein, MCT8 [1]. The disease was first described in 1944 and was among the first of the X-linked syndromes associated with intellectual disability [2]. AHDS results from a mutation of the SLC16A2 gene on the X chromosome leading to a deficiency of the MCT8 protein. MCT8 creates a channel allowing the thyroid hormone, triiodothyronine (T3) to move from the peripheral blood stream into the CNS [3-7]. As there is a central lack of T3, increased production of thyroid-stimulating hormone (TSH) results in the peripheral overproduction of T3 resulting in systemic signs and symptoms of hyperthyroidism, while central lack of T3 results in severe CNS effects including neurodevelopment delay, intellectual disability, hypotonia, and failure to thrive [8]. We present a 6-year-old child with AHDS who required anesthetic care during various anesthetic encounters including right open adductor tenotomy, right proximal femoral varus osteotomy, and right acetabular osteotomy. The basic cellular defect of AHDS is described, end-organ effects outlined, and impact on perioperative care reviewed. This review was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
| Case Report | ▴Top |
Review of this case and presentation in this format followed the guidelines of the Institutional Review Board of Nationwide Children’s Hospital. The patient was a 6-year-old, 30.2 kg male with ADHS, who presented for anesthetic care for multiple procedures during the first 5 - 6 years of life including interventional radiology for liver biopsy, bilateral tympanostomy tube insertion, esophagogastroduodenoscopy (EGD) with placement of percutaneous endoscopic gastrostomy (PEG) tube insertion, radiologic imaging, and a recent orthopedic surgical procedure (right open adductor tenotomy, right proximal femoral varus osteotomy, and right acetabular osteotomy). Associated comorbid conditions included failure to thrive, developmental delay, spastic quadriplegic cerebral palsy, hypotonia, dysphagia with aspiration, hypothyroidism, seizures, and asthma. Current medications included levetiracetam and clobazam oral suspensions administered through the gastrostomy.
The child was born at term via spontaneous vaginal delivery without any perinatal complications. At approximately 3 months of age, he was evaluated for failure to thrive and noted to have abnormal thyroid function tests consistent with hypothyroidism. Oral thyroxine administration was started. Additional evaluation was initiated for failure to thrive, hypotonia, and neutropenia during the time period from 3 to 7 months of age by various subspecialty services including hematology, neurology, and genetics. Although the single nucleotide polymorphisms (SNPs) genotyping analysis at 7 - 8 months of age was normal, a more specific Region of Interest Panel (Claritas Genomics) identified the MCT-specific thyroid hormone cell-membrane transporter deficiency and the diagnosis of AHDS was made after exome sequencing slice showing a hemizygous pathogenic variant in SLC16A2 (X-linked MCT8-specific thyroid hormone cell membrane transport protein deficiency). Family history was significant for intellectual disability and short stature in the mother as well as intellectual disability in a maternal uncle and grandmother.
The first anesthetic encounter was at 4 months of age for magnetic resonance (MR) imaging (patient weight 4.9 kg). Anesthesia was induced by the inhalation of sevoflurane in nitrous oxide and oxygen. Tracheal intubation was facilitated by the administration of cis-atracurium, and maintenance anesthesia included sevoflurane in air and oxygen. At the completion of the procedure, residual neuromuscular blockade was reversed with neostigmine. The postoperative course was uneventful. The second anesthetic encounter was at 2 years of age after the diagnosis of AHDS was established. The child weighed 9.85 kg and presented to interventional radiology for a liver biopsy to evaluate persistently elevated liver function tests. The patient’s preoperative vital signs were within the normal range for age. A thyroid profile was performed 3 months prior (TSH 10.155 IU/mL, free T4 0.6 ng/dL). Anesthesia was induced by the inhalation of sevoflurane in air and oxygen. Tracheal intubation was facilitated by the administration of propofol (about 5 mg/kg) and fentanyl (about 2 µg/kg). The liver biopsy was completed without incident and the postoperative course was uncomplicated. At 3 years of age (patient weight 12.4 kg), the patient presented for anesthetic care during examination under anesthesia and placement of bilateral tympanostomy tubes. Anesthesia was induced by the administration of incremental concentrations of sevoflurane in nitrous oxide and oxygen. Maintenance anesthesia included sevoflurane in air and oxygen with spontaneous ventilation. Intranasal fentanyl (2 µg/kg) was administered for postoperative analgesia. The intraoperative and postoperative courses were uneventful, and the patient was discharged home the same day. Preoperative thyroid testing revealed: TSH 12.60 IU/mL, free T4 0.6 ng/dL, total T3 290 ng/dL). Postoperative results were: TSH 10.30 IU/mL, free T4 0.6 ng/dL, and total T3 284 ng/dL. The following year, the patient required anesthesia for MR imaging to evaluate new onset seizures. At the time, the patient’s weight remained stable at 12.8 kg. Anesthesia was induced by the administration of incremental concentrations of sevoflurane in nitrous oxide and oxygen. Propofol (40 mg) was administered to facilitate placement of a laryngeal mask airway (LMA). Maintenance anesthesia included sevoflurane in air and oxygen with spontaneous ventilation through the LMA. Dexamethasone and ondansetron were administered to provide prophylaxis against postoperative nausea and vomiting (PONV). The intraoperative and postoperative course were unremarkable. At 5 years of age, the patient presented for anesthetic care during EGD with PEG insertion. The patient weighed 19.3 kg. Preoperative vital signs were within normal ranges for age. Anesthesia was induced by the administration of incremental concentrations of sevoflurane in nitrous oxide and oxygen. Endotracheal intubation was facilitated by the administration of propofol (2 mg/kg). Anesthesia was maintained with sevoflurane in oxygen. During EGD, it was determined that the stomach was positioned underneath the left ribcage, making it difficult to place the PEG tube, and G-tube placement was not successful. PONV prophylaxis was provided with dexamethasone and ondansetron. The postoperative course was uneventful, and the patient was scheduled to return for surgical G-tube placement. A few months later, the patient presented for anesthetic care during laparoscopic G-tube placement. At this time, he weighed 22.7 kg. Anesthesia was induced by the administration of incremental concentrations of sevoflurane in nitrous oxide and oxygen. After the induction of anesthesia, a peripheral intravenous cannula was placed and rocuronium (20 mg) was administered to facilitate endotracheal intubation and provide neuromuscular blockade for the surgical procedure. Maintenance anesthesia included sevoflurane in air and oxygen supplemented with dexmedetomidine (0.5 µg/kg). Morphine (about 0.1 mg/kg) and acetaminophen were administered for postoperative analgesia, and ondansetron and dexamethasone for prophylaxis against PONV. An additional surgical intervention was planned after G-tube placement and improvement of his nutritional status to treat right hip dislocation secondary to neuromuscular hip dysplasia.
At 6 years of age (24.8 kg), surgical intervention to treat right hip subluxation was scheduled including open adductor tenotomy, right proximal femoral varus osteotomy, and right acetabular osteotomy. The patient was noted to have tachycardia (120 - 130 beats/min) at baseline with an oxygen saturation of 93% on room air. The family denied any recent illness but did note that he was having seizures several times per day. A thyroid function test was performed approximately 1 year prior to this surgical procedure (TSH 4.671 IU/mL, free T4 0.6 ng/dL). The family also reported a history of oxygen desaturation following anesthesia due to agitation. He was held appropriately nil per os (NPO) and was transported to the operating room where routine American Society of Anesthesiologists’ monitors were applied. Anesthesia was induced by the inhalation of incremental concentrations of sevoflurane in nitrous oxide and oxygen. Following the induction of anesthesia, a peripheral intravenous cannula was placed. Neuromuscular blockade was provided by rocuronium (1 mg/kg) and anesthesia supplemented with propofol (1 mg/kg). Following direct laryngoscopy with a Macintosh blade 2, the trachea was intubated with a 5.0-mm cuffed endotracheal tube (ETT). Anesthesia was maintained with sevoflurane in air and oxygen and fentanyl (2 µg/kg). A right-sided suprainguinal fascia iliaca block was performed with 20 mL of 0.2% ropivacaine with epinephrine (1:200,000) and dexamethasone (0.2 mg/mL). Surgical site prophylaxis against infection was provided with cefazolin (50 mg/kg). The intraoperative course was unremarkable. Additional postoperative analgesia was provided by hydromorphone (0.012 mg/kg) and acetaminophen. Prophylaxis against PONV included dexamethasone and ondansetron. At the completion of the surgical procedure, residual neuromuscular blockade was reversed with sugammadex (2 mg/kg), the patient’s trachea was extubated awake, and he was transported to the post-anesthesia care unit (PACU). In the PACU, the patient became increasingly somnolent and difficult to arouse, and two doses of naloxone (1 µg/kg/dose) were administered. Due to respiratory concerns, the pain team and complex care team were consulted, and the patient was admitted to the inpatient ward with continuous oxygen saturation monitoring. Transfer to the pediatric ICU was required later that evening for increased postoperative monitoring and respiratory support. A postoperative chest radiograph, obtained due to a persistent oxygen requirement and low oxygen saturation, revealed left lower lobe atelectasis. Aggressive chest physiotherapy and suctioning was provided to treat mucus plugging and sialorrhea. The latter was also treated with sublingual atropine drops. Parents reported that secretions and frequent oxygen desaturations had been an ongoing issue at home requiring frequent deep suctioning and vest physiotherapy. Consideration was being given to salivary gland ablation in the future. The following day, the patient was transferred to the inpatient ward. Thyroid function tests were repeated on postoperative day 1 (TSH 2.370 IU/mL, free T4 0.9 ng/dL, total T3 193 ng/dL, free T3 4.9 pg/mL). The remainder of his postoperative course was unremarkable, and he was discharged home on postoperative day 2.
| Discussion | ▴Top |
AHDS is an X-linked disorder characterized by hypotonia, feeding difficulties, developmental delay, and later onset of dystonia, hypokinesia, and choreoathetosis. The pathophysiology involves dysfunction of the MCT8 protein, which is responsible for transport of T3 into the CNS. Reduced T3 in the CNS leads to impairment of neurogenesis, defective migration and myelination which manifests as neurological, developmental, and psychomotor disabilities. Due to decreased central T3, there is up-regulation of TSH release with peripheral signs of hyperthyroidism. Treatment with thyroxine replacement therapy (levothyroxine) remains ineffective against central and peripheral thyroid derangement due to defective transport of the exogenous hormone into the CNS due to deficiency of the MCT8 transport protein. Alternative therapies, diiodothyropropionic acid (DITPA), an agonist of T3 within the CNS, and triiodothyroacetic acid, a thyroid hormone analog that can enter cells via alternative transport mechanisms that do not rely on the MCT8 transporter, are being investigated.
As noted in our patient, anesthetic care is frequently required for diagnostic imaging and for surgical procedures to palliate or treat end-organ involvement related to AHDS. Given the multi-system involvement of AHDS, several perioperative concerns may be encountered. A thorough preoperative history and physical examination to identify comorbid conditions related to the primary disease process should be completed and the patient’s status optimized prior to anesthesia. Because of the complexity of these patients, their varied clinical picture, and changing medication regimens, a key component of the preoperative evaluation should be an inclusive review of the current and recent medication record, thyroid function tests, and vital signs. Communication and collaboration with the patient’s primary care physicians including endocrinology and neurology should be sought as needed. Patients with known genetic syndromes pose a variety of challenges to the anesthesia provider including the potential for difficulties with airway management, bag-valve-mask ventilation or endotracheal intubation [9-11]. As noted in our patient, the preoperative evaluation and care of these patients may be compounded by the rarity of the disorder and lack of previous reports in the literature regarding anesthetic care; however, specific airway concerns have not been reported in patients with AHDS. Our patient’s preoperative airway evaluation was unremarkable and failed to reveal anatomical findings which may suggest problematic airway management [10]. Additionally, prior anesthetic care revealed no history of difficulties with airway management. However, the appropriate equipment for dealing with the difficult airway should be readily available prior to anesthetic induction and adequate bag-valve-mask ventilation demonstrated prior to the administration of a neuromuscular blocking agent [10, 11]. As was performed in our patient, general anesthesia can be induced by the incremental inhalation of sevoflurane with the maintenance of spontaneous ventilation until effective bag-valve-mask ventilation is demonstrated. Once effective bag-valve-mask ventilation was demonstrated, the depth of anesthesia was deepened with the administration of propofol followed by endotracheal intubation. In patients with CNS disabilities, airway management may be further compromised by the risk of aspiration due to gastroesophageal reflux (GER) which may necessitate a rapid sequence intubation (RSI). The use of RSI must be weighed against the risks of potential difficulties with airway management and endotracheal intubation. In our patient, there were no specific clinical indications of GER; however, he was noted to have difficulty with excessive oral secretions.
Hypotonia, which is a frequent comorbid condition of various syndromes, including AHDS, may affect perioperative care and impact choice and subsequent use of neuromuscular blocking agents (NMBAs) [12, 13]. Depending on the underlying etiology of hypotonia and the associated involvement of the skeletal muscle and neuromuscular junction, succinylcholine may be contraindicated, given the potential for rhabdomyolysis and hyperkalemia [14]. Given the lack of previous clinical reports and paucity of clinical experience with AHDS patients, no definitive recommendations can be given regarding the safety of succinylcholine. When considering the patient’s clinical status, comorbid involvement, and the anticipated surgical procedures, there were several options chosen during the anesthetic care of our patient. For procedures not requiring neuromuscular blockade, endotracheal intubation can be performed with deep inhalational anesthesia with sevoflurane, supplemented as needed by the administration of propofol and/or opioids (fentanyl or remifentanil). Alternatively, diagnostic imaging can frequently be performed without endotracheal intubation and placement of an LMA, which does not require neuromuscular blockade. For longer procedures, we chose to use rocuronium to facilitate endotracheal intubation and provide neuromuscular blockade during the procedure (G-tube placement). In our anecdotal experience, despite the associated CNS involvement and hypotonia, no prolongation of the effect of rocuronium was noted. However, non-depolarizing NMBAs should be used with care as the effect can be prolonged even with routine dosing in patients with pre-existing neuromuscular diseases or hypotonia [12, 13]. When available, sugammadex may provide an additional margin of safety for reversal of the neuromuscular blocking effects of rocuronium or vecuronium, especially in patients with hypotonia or neuromuscular disorders [15-17].
Perioperative care may also be impacted by poor airway control, hypotonia, and respiratory muscle insufficiency in patients with CNS disabilities resulting in obstructive sleep apnea (OSA), sensitivity to opioids and residual anesthetic agents. As demonstrated by our patient, various factors may combine to result in postoperative hypoventilation, atelectasis, poor airway clearance, mucus plugging, and the risk for perioperative respiratory insufficiency. In our patient, it is likely that the combined effects of the comorbid involvement of AHDS with hypotonia, poor cough effort, mucus plugging, and sensitivity to the respiratory depressant effects of opioids resulted in hypoventilation and atelectasis. Emergence from anesthesia in our patient was prolonged, and two doses of naloxone were administered to reverse the effects of intraoperative opioids. Although we initially planned on care on the inpatient ward, progressive hypoxemia and oxygen desaturation necessitated transfer to the pediatric ICU for closer monitoring and more aggressive chest physiotherapy. Although our patient had previously tolerated opioid medications well, the efficacy of the regional block combined with progression of the underlying disease process likely contributed to a lower requirement overall. Previous experience has demonstrated the utility of regional blockade and non-opioid adjuncts such as acetaminophen in limiting the need for postoperative opioids and their impact on respiratory function in patients with CNS disabilities. The potential for perioperative respiratory compromise mandates continuous respiratory monitoring (pulse oximetry) in these patients. Progressive atelectasis and mucus plugging that fails to respond to aggressive chest physiotherapy may require the application of bilevel positive airway pressure (BiPAP) to prevent the need for tracheal intubation.
Patients with AHDS exhibit additional neurological involvement including spasticity and oropharyngeal involvement [18, 19]. These neurological impairments can affect the coordination and function of the muscles involved in swallowing, secretion management, and maintenance of airway patency. Oropharyngeal dysfunction can impair the ability to swallow effectively, leading to difficulties in clearing oropharyngeal secretions which may further impact postoperative respiratory function. Increased secretions were noted perioperatively in our patient, requiring the addition of pharmacologic management (sublingual atropine drops). Airway patency may be further compromised by associated macroglossia [18]. These factors contribute to the development of sleep disordered breathing and OSA, which may be exacerbated during the postoperative period [20].
Extrapyramidal symptoms are also a part of the clinical picture and may be evident from an early age including dystonic posturing of the upper limbs, fluctuating muscle tone, and an increased startle reaction. The inability to walk is a prominent feature associated with dystonia, seen in a majority of patients along with other extrapyramidal symptoms and movement disorders (choreoathetosis). Intellectual disability is uniformly present ranging from mild/moderate to severe/profound developmental delay and cognitive impairment.
Joint and soft tissue contractures may lead to problems with intraoperative positioning. This may be further impacted by orthopedic deformities, including kyphoscoliosis, and poor nutritional status. Close attention to positioning and appropriate padding of bony prominences are imperative to prevent skin breakdown during prolonged procedures or immobilization. Growth retardation, nutritional issues, and CNS disabilities may predispose to temperature instability with hypothermia, especially during prolonged surgical procedures. Thus, care must be taken to reduce the risk of intraoperative hypothermia with continuous intraoperative temperature monitoring and the use of overhead heating lights, forced air warming devices, increased room temperature, and heated and humidified inspired gases.
Learning points
AHDS is a rare X-linked disorder resulting from mutation of the SLC16A2 gene leading to a deficiency of a protein which facilitates the passage of T3 into the CNS. Affected individuals will have an elevated free T3 concentration, low free T4, normal-elevated levels of TSH, and a free T3/T4 ratio greater than 0.75. This occurs despite central hypothyroidism because MCT8 is essential for the entry of thyroid hormones into the CNS. Unfortunately, due to the role of thyroid hormones during the early in utero development of the brain, affected patients have profound impairment of development and significant cognitive impairment. Various other end-organ involvement has been noted which may impact perioperative care (Table 1) during diagnostic imaging or surgical procedures to treat or palliate sequelae of the disease process. Although there are anecdotal reports of patients surviving into the fifth or sixth decade of life, overall life expectancy is significantly compromised, and the quality of life severely affected as most patients are unable to walk independently or feed themselves. Throughout their lifetime patients with AHDS are likely to require multiple anesthetics and require careful preoperative evaluation and a perioperative plan tailored to their specific comorbidities. Although there are no specific anesthetic medications known to be contraindicated in patients with AHDS, natural progression of the disease process may lead to increased sensitivity to anesthetic agents, NMBAs, and opioid medications over time, which must be considered with each subsequent anesthetic encounter.
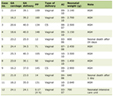 Click to view | Table 1. End-Organ Involvement and Perioperative Concerns of Patients With AHDS |
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
Following the guidelines of the IRB of Nationwide Children’s Hospital (Columbus, Ohio), informed consent was obtained for anesthetic care and the use of deidentified information for publication.
Author Contributions
Preparation of initial, subsequent, and final drafts (WNK); concept, writing, and review of all drafts (JDT); perioperative care of patient, review of final draft (RH).
Data Availability
Any inquiries regarding supporting data availability of this study should be directed to the corresponding author.
| References | ▴Top |
- Schwartz CE, May MM, Carpenter NJ, Rogers RC, Martin J, Bialer MG, Ward J, et al. Allan-Herndon-Dudley syndrome and the monocarboxylate transporter 8 (MCT8) gene. Am J Hum Genet. 2005;77(1):41-53.
doi pubmed pmc - Allan W, Herndon CN, Dudley FC. Some examples of the inheritance of mental deficiency: apparently sex-linked idiocy and microcephaly. Am J Ment Defic. 1944;48:325-334.
- Visser WE, Vrijmoeth P, Visser FE, Arts WF, van Toor H, Visser TJ. Identification, functional analysis, prevalence and treatment of monocarboxylate transporter 8 (MCT8) mutations in a cohort of adult patients with mental retardation. Clin Endocrinol (Oxf). 2013;78(2):310-315.
doi pubmed - Heuer H, Maier MK, Iden S, Mittag J, Friesema EC, Visser TJ, Bauer K. The monocarboxylate transporter 8 linked to human psychomotor retardation is highly expressed in thyroid hormone-sensitive neuron populations. Endocrinology. 2005;146(4):1701-1706.
doi pubmed - Ceballos A, Belinchon MM, Sanchez-Mendoza E, Grijota-Martinez C, Dumitrescu AM, Refetoff S, Morte B, et al. Importance of monocarboxylate transporter 8 for the blood-brain barrier-dependent availability of 3,5,3'-triiodo-L-thyronine. Endocrinology. 2009;150(5):2491-2496.
doi pubmed pmc - Felmlee MA, Jones RS, Rodriguez-Cruz V, Follman KE, Morris ME. Monocarboxylate Transporters (SLC16): Function, Regulation, and Role in Health and Disease. Pharmacol Rev. 2020;72(2):466-485.
doi pubmed pmc - Boccone L, Dessi V, Meloni A, Loudianos G. Allan-Herndon-Dudley syndrome (AHDS) in two consecutive generations caused by a missense MCT8 gene mutation. Phenotypic variability with the presence of normal serum T3 levels. Eur J Med Genet. 2013;56(4):207-210.
doi pubmed - Mullur R, Liu YY, Brent GA. Thyroid hormone regulation of metabolism. Physiol Rev. 2014;94(2):355-382.
doi pubmed pmc - Butler MG, Hayes BG, Hathaway MM, Begleiter ML. Specific genetic diseases at risk for sedation/anesthesia complications. Anesth Analg. 2000;91(4):837-855.
doi pubmed pmc - Bryant J, Krishna SG, Tobias JD. The difficult airway in pediatrics. Advan Anesth. 2013;31:31-60.
- Engelhardt T, Weiss M. A child with a difficult airway: what do I do next? Curr Opin Anaesthesiol. 2012;25(3):326-332.
doi pubmed - Azar I. The response of patients with neuromuscular disorders to muscle relaxants: a review. Anesthesiology. 1984;61(2):173-187.
doi pubmed - Ragoonanan V, Russell W. Anaesthesia for children with neuromuscular disease. Contin Educ Anaesth Crit Care Pain. 2010;10:143-147.
- Martyn JA, Richtsfeld M. Succinylcholine-induced hyperkalemia in acquired pathologic states: etiologic factors and molecular mechanisms. Anesthesiology. 2006;104(1):158-169.
doi pubmed - Pani N, Dongare PA, Mishra RK. Reversal agents in anaesthesia and critical care. Indian J Anaesth. 2015;59(10):664-669.
doi pubmed pmc - Dontukurthy S, Wisler C, Raman V, Tobias JD. Myasthenia gravis and sugammadex: A case report and review of the literature. Saudi J Anaesth. 2020;14(2):244-248.
doi pubmed pmc - Tobias JD. Current evidence for the use of sugammadex in children. Paediatr Anaesth. 2017;27(2):118-125.
doi pubmed - Remerand G, Boespflug-Tanguy O, Tonduti D, Touraine R, Rodriguez D, Curie A, Perreton N, et al. Expanding the phenotypic spectrum of Allan-Herndon-Dudley syndrome in patients with SLC16A2 mutations. Dev Med Child Neurol. 2019;61(12):1439-1447.
doi pubmed - Tonduti D, Vanderver A, Berardinelli A, Schmidt JL, Collins CD, Novara F, Genni AD, et al. MCT8 deficiency: extrapyramidal symptoms and delayed myelination as prominent features. J Child Neurol. 2013;28(6):795-800.
doi pubmed pmc - Ankichetty S, Wong J, Chung F. A systematic review of the effects of sedatives and anesthetics in patients with obstructive sleep apnea. J Anaesthesiol Clin Pharmacol. 2011;27(4):447-458.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.
