| International Journal of Clinical Pediatrics, ISSN 1927-1255 print, 1927-1263 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Int J Clin Pediatr and Elmer Press Inc |
| Journal website https://www.theijcp.org |
Case Report
Volume 11, Number 1, March 2022, pages 27-32
Hemolysis in Neonates Born to Mothers With History of SARS-CoV-2 Infection During Pregnancy: A Case Series
Elena Itriagoa, Krithika Lingappanb, Jun Teruyac, Daniela Dinua, d
aDepartment of Pediatrics, Section of Neonatology, Baylor College of Medicine, Houston, TX, USA
bDivision of Neonatology, The Children’s Hospital of Philadelphia, Philadelphia, PA, USA
cDepartment of Pathology & Immunology, Baylor College of Medicine, Houston, TX, USA
dCorresponding Author: Daniela Dinu, Department of Pediatrics, Section of Neonatology, Baylor College of Medicine, Houston, TX, USA
Manuscript submitted February 7, 2022, accepted February 28, 2022, published online March 12, 2022
Short title: Hemolysis in Preterms Born to SARS-CoV-2 Mothers
doi: https://doi.org/10.14740/ijcp479
| Abstract | ▴Top |
Hemolytic anemia has been associated with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in adult and pediatric patients. Here we report a case series of preterm newborns with hemolytic anemia whose mothers had a history of SARS-CoV-2 infection. In two of the cases maternal infection occurred early in pregnancy and the newborns had a reactive SARS-CoV-2 antibody test. Hemolysis and coagulopathy improved with the use of washed blood products and prothrombin complex concentrate. Our cases highlight the need for increased awareness of a possible association between hemolysis and exposure to SARS-CoV-2 infection during intrauterine life.
Keywords: Hemolysis; SARS-CoV-2; Neonate; Pregnancy
| Introduction | ▴Top |
While respiratory symptoms are the most common, extra-pulmonary manifestations of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in adults are becoming increasingly described, with both Coombs-positive and Coombs-negative hemolytic anemia reported in the literature [1-3]. However, the precise mechanism that leads to hemolysis in patients with SARS-CoV-2 infections is still not completely understood [4].
Studies looking at the effects of SARS-CoV-2 infection on the placenta and the newborn have indicated a possible increased risk of preterm birth [5], while others have reported respiratory distress, tachycardia, shock, thrombocytopenia, and even death or stillbirth in some neonates born to mothers with coronavirus disease 2019 (COVID-19) [6, 7]; and a recent meta-analysis concluded that maternal SARS-CoV-2 infection in pregnancy was significantly associated with small increases in some neonatal morbidities such as: admission for neonatal care, neonatal respiratory disorders and hyperbilirubinemia [8].
Vertical transmission of SARS-CoV-2 while possible, remains rare and most neonates born to mothers with SARS-CoV-2 infection during pregnancy test negative for SARS-CoV-2 [9]; but newborns born to both symptomatic and asymptomatic SARS-CoV-2-positive mothers have detectable levels of immunoglobulin G (IgG) antibodies transferred across the placenta, and these antibodies are believed to have protective effect for the newborns [10, 11]. It is also postulated that in certain genetically susceptible newborns, these antibodies may bind to receptors on neutrophils and macrophages causing activation of cytokines leading to various manifestations consistent with neonatal multisystem inflammatory syndrome [12].
Here, we describe three neonates born to mothers with history of SARS-CoV-2 infection during pregnancy who developed hemolytic anemia.
| Case Reports | ▴Top |
Case 1
A baby girl of 29 1/7 gestational weeks born to a 22 years old primigravida mother with SARS-CoV-2 pneumonia, acute respiratory failure and shock (Table 1) was delivered due to non-reassuring fetal heart tones with prolonged decelerations and sinusoidal pattern. There was no history of smoking or substance abuse. Maternal history was significant for COVID pneumonia and respiratory failure requiring intubation and transfer to our hospital, followed 24 h later by transfer to another facility for extracorporeal membrane oxygenation cardio-respiratory support. During admission, maternal medications included: ceftriaxone, azithromycin, norepinephrine, vasopressin, propofol, midazolam, fentanyl, cisatracurium and hydroxychloroquine, At the delivery baby’s Apgar score were 1, 3 and 7 at 1, 5 and 10 min, respectively, and required positive pressure ventilation followed by endotracheal intubation for apnea and high oxygen requirement. She received one dose of surfactant and was admitted to neonatal intensive care unit (NICU). On admission, her temperature was 37.8 °C with a pulse of 150/min, respiratory rate of 50/min, oxygen saturation of 96% on 40% oxygen, and blood pressure (BP) of 48/20 mm Hg with a mean of 29 mm Hg. Birth weight was 1,275 g (64.7th percentile), length 38 cm (61.6th percentile) and head circumference 26.3 cm (54.3th percentile). A limited sepsis evaluation was performed, and she received intravenous ampicillin and gentamicin for 48 h until blood culture was negative. On physical exam she was alert and active, head normocephalic with anterior fontanel open and flat, no dysmorphic features were appreciated, lungs had good air entry bilaterally, normal sinus rhythm, no murmurs, abdomen soft, not distended, no organomegaly, anus patent, symmetrical Moro and decreased tone as expected for her gestational age. Her respiratory status improved, and she was extubated to continuous positive pressure on first day of life (DOL). She remained on continuous positive airway pressure (CPAP), and she tolerated full enteral feeds with donor breast milk and human milk derived fortifier until her episode of clinical deterioration. On DOL 17 she developed abdominal distension, hypotension, and bloody stools. She underwent another sepsis evaluation, and antibiotics (vancomycin and gentamicin) were started. The initial abdominal X-ray was negative for pneumatosis, and an abdominal ultrasound was negative for volvulus or malrotation. She received red blood cells (RBCs) transfusion for hemoglobin of 10.1 g/dL. On DOL 18, her status deteriorated with respiratory failure requiring intubation, refractory hypotension requiring vasopressors, persistent bloody stools, and hemoglobinuria. A blood transfusion reaction was suspected, and given minor crossmatch positive, T-cryptantigen (TCA) activation was considered likely. The peripheral blood smear reported anemia with moderate anisocytosis, nucleated RBC, and increased spherocytes and schistocytes. Plasma free hemoglobin was elevated to 2,280 mg/dL. She also developed thrombocytopenia and coagulopathy (Table 2). The patient received blood products that were less likely to exacerbate hemolysis: washed RBC and platelets, prothrombin complex concentrate (PCC), and fibrinogen concentrate with improvement in coagulopathy.
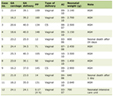 Click to view | Table 1. Maternal Severity of COVID Disease, Birth History and Newborn Anthropometric Data |
 Click to view | Table 2. Laboratory Results |
The clinical course worsened with multiorgan failure (metabolic acidosis, acute kidney injury) with continued abdominal distension and coagulopathy. Due to concern for possible ongoing bowel ischemia or perforation she underwent an exploratory laparotomy where a large sigmoid perforation was found. On DOL 19 the abdominal distension worsened and was accompanied by discoloration of the overlying skin. The coagulopathy improved after receiving multiple doses of washed platelets and PCC. She continued to be hypotensive requiring vasopressors, with persistent metabolic acidosis and increasing lactate. Furthermore, secondary to severe kidney dysfunction with anuria she developed more symptomatic hyperkalemia and hypocalcemia with arrhythmias. Over the next 2 days, the baby underwent two additional explorations of the abdomen and entire small bowel, and colon were found to be ischemic, with multiple focal areas of necrosis. After discussions with the family, the care was redirected.
The autopsy’s report was consistent with necrotizing enterocolitis (NEC) totalis, diffuse hypoxic-ischemic injury in the cerebral cortex, basal ganglia and thalamus, hippocampus and brainstem and extensive subarachnoid hemorrhage. Additionally, multiorgan microvascular fibrin thrombi involving heart, renal glomeruli, stomach, small and large bowel may have been associated with intravascular hemolysis.
During this episode of decompensation, she received multiple medications in addition to vancomycin and gentamicin including: calcium gluconate continuous infusion and boluses, dexamethasone, dopamine, fentanyl, hydrocortisone, insulin, albuterol, piperacillin-tazobactam, sodium bicarbonate infusion and boluses, vasopressin.
Case 2
A preterm boy of 27 5/7 gestational weeks was born to a 31 years old primigravida mother by cesarean section due to reversed end-diastolic flow and severe intrauterine growth restriction (IUGR). There was a history of fetal growth restriction diagnosed on the 23 weeks anatomy scan with an estimated fetal weight < 1st percentile. Pregnancy was complicated by maternal history of systemic lupus erythematosus, Crohn’s disease, and asymptomatic maternal SARS-CoV-2 infection 2 months before delivery. There was no history of alcohol use or smoking. Maternal medications included: enoxaparin prophylaxis for antiphospholipid syndrome, methylprednisolone pulse for 3 days prior to delivery followed by oral prednisone, azathioprine, hydroxychloroquine, as well as betamethasone for fetal lung maturation. She was group B streptococcus positive; however, no antibiotics were administered due to delivery for fetal indication in the absence of labor. Apgar scores were 4 and 8, and he required positive pressure ventilation in the delivery room followed by CPAP. On admission to NICU, his vitals were: temperature 37.2 °C, pulse 150/min, respiratory rate 35/min, BP 63/29 mm Hg with a mean of 41 mm Hg, oxygen saturation 94% on 30% oxygen. His weight was 665 g (5.1th percentile), length 31 cm (1.7th percentile) and head circumference 23.6 cm (10.6th percentile). On physical exam he was active, well hydrated, head normocephalic, anterior fontanelle open and flat, no dysmorphic features, had diminished breath sounds bilaterally, normal sinus rhythm with no murmurs, abdomen soft, not distended, no organomegaly, normal male genitalia; he was moving all extremities well and there were no focal deficits. He was started on caffeine and vitamin A for bronchopulmonary dysplasia prophylaxis based on his birth weight as per our unit protocol. On DOL 1, he was found to be pancytopenic, followed on DOL 2 by mild disseminated intravascular coagulation (DIC), which responded to plasma, RBC, and platelet transfusions. On DOL 2, due to worsening thrombocytopenia a limited sepsis evaluation was performed, and he received ampicillin and amikacin for 48 h until blood culture was negative. The patient had evidence of intravascular hemolysis with a hemoglobin of 7.2 g/dL, elevated unconjugated bilirubin (7 mg/dL), lactate dehydrogenase (LDH) of 555 U/L, high reticulocyte count, and plasma free hemoglobin (700 mg/dL); the peripheral smear reported nucleated RBC and schistocytes. The lupus anticoagulant panel was negative. Head ultrasound reported no intraventricular hemorrhage. SARS-CoV-2 antibodies were reactive (Table 2). He remained on CPAP until DOL 52 when he was weaned to high-flow nasal cannula, and then room air at a postmenstrual age of 36 5/7 weeks. The medications administered during his hospital stay included caffeine, furosemide for 3 days and two short courses of antibiotics for presumed sepsis. He was discharged home on DOL 84, at a postmenstrual age of 39 5/7 weeks. Of note, his newborn screen was concerning for possible severe combined immunodeficiency, and he followed as an outpatient with the immunology service. He was diagnosed with mild T cell lymphopenia of his CD3/4/8 cells which resolved by 1 year of age, and was deemed secondary to prematurity, as well as transient hypogammaglobulinemia of infancy and neutropenia, but with adequate absolute neutrophil count and no episodes of infections.
Case 3
A preterm boy of 36 gestational weeks was delivered to a 27 years old primigravida mother by cesarean section due to fetal intolerance to labor manifested as prolonged decelerations to 90/min with a baseline fetal heart rate of 150/min, which recovered only partially with repositioning. His Apgar scores were 8 and 9, and he required brief oxygen administration for hypoxemia. Mother was found to be group B streptococcus positive and received vancomycin, due to history of penicillin allergy, 6 h prior to delivery. Pregnancy was complicated by SARS-CoV-2 infection during the second trimester, type 2 diabetes mellitus, chronic hypertension, oligohydramnios, and chorioamniotic separation. Maternal symptoms related to COVID infection were cough and vomiting, and she did not require treatment or hospital admission. Maternal medications during pregnancy included labetalol and insulin, and there was no reported history of smoking or alcohol use during pregnancy. At birth, the newborn had a glucose level of 30 mg/dL, and he received dextrose gel and oral formula, and repeat glucose was again 30 mg/dL which prompted NICU admission. Upon NICU admission, his temperature was 36.8 °C, heart rate 142/min, BP 74/53 mm Hg, respirations 66/min, and oxygen saturation 95% on room air, birth weight 2,800 g (53.5th percentile), length 47 cm (44.3th percentile) and head circumference 31 cm (11.8th percentile). On physical exam he was well appearing, pink and vigorous, normocephalic, anterior fontanelle open and flat, with no dysmorphic features, breath sounds clear and equal bilaterally, normal sinus rhythm with no murmurs, abdomen soft with no masses and no organomegaly, anus patent, normal male genitalia, normal muscle tone, Moro reflex symmetrical, rooting, sucking and grasp reflexes present and no focal deficits. At 24 h of life, he was found to have elevated total bilirubin of 13.6 mg/dL requiring phototherapy and close to the threshold needing exchange transfusion. Additional investigations were consistent with intravascular hemolysis with elevated reticulocyte count, elevated immature reticulocyte fraction, elevated free plasma hemoglobin (120 mg/dL), elevated LDH (1,853 U/L) and low haptoglobin (< 8 mg/dL). SARS-CoV-2 antibodies were reactive (Table 2). Hyperbilirubinemia responded to intensive phototherapy. Glucose-6 phosphate dehydrogenase (G6PD) level was low at 5.4 U/g hemoglobin. During his hospital stay he received ampicillin and amikacin for 48 h as part of sepsis evaluation. He was discharged from the hospital on DOL 8 and was seen in the Hematology Clinic at 6 weeks of life, as well as being followed regularly by his pediatrician. He has remained clinically well, with no additional concerns for hemolysis and no further investigations.
| Discussion | ▴Top |
We describe the clinical course of three neonates born to mothers with history of SARS-CoV-2 infection during pregnancy, who developed intravascular hemolysis while having other concurrent illnesses. No SARS-CoV-2 vaccine was available at the time of hospital admission, and all cases were diagnosed using SARS-CoV-2 polymerase chain reaction (PCR) of nasopharyngeal swabs, as well as symptoms consistent with COVID infection for mothers of patients 1 and 3. Two cases had a reactive anti-SARS-CoV-2 antibodies test. Case 1 had two negative nasopharyngeal swabs for SARS-CoV-2 PCR; cases 2 and 3 were not tested given the negative maternal tests at the delivery. Vertical transmission of SARS-CoV-2, although rare, has been reported, but our cases did not meet the criteria for congenital infection [13]. However, there have been reports of newborns born to SARS-CoV-2-positive mother who presented with thrombocytopenia, refractory shock, and multiorgan failure [14]. Another study reported NEC in a preterm infant born to a SARS-CoV-2-positive mother [15]. All our cases developed hemolytic anemia early in their course, and it was associated with conditions that predispose to hemolysis such as NEC, G6PD deficiency, and DIC.
Our first case had NEC, a complication of prematurity with an incidence of 5-10% and with mortality rates as high as 30% [16, 17]. Anemia is well described in NEC and is multi-factorial due to bleeding, iatrogenic losses, thrombotic microangiopathy, or activation of the Thomsen-Friedenrich cryptantigen on the RBC. As many as 30% of newborns with NEC could have TCA activation [18]. Surface TCA is an antigen concealed by a layer of N-acetylneuraminic acid. When exposed, TCA may interact with IgM anti-T antibodies present in plasma [19]. When RBCs are destroyed, hemoglobin is released in plasma and couples irreversibly with haptoglobin. This complex is cleared from circulation by monocytes and macrophages. Low levels of haptoglobin, as in cases 2 and 3, are useful to support the hemolytic etiology, but it is important to note that solely low haptoglobin levels in newborns may not be useful for diagnosis of hemolysis [20]. In case 1, there was a strong clinical suspicion that TCA activation was responsible for the hemolytic process based on positive minor crossmatch. The patient received washed RBC and platelets, PCC, and fibrinogen concentrate with subsequent improvement in hemolysis. Case 3 had decreased level of G6PD. G6PD deficiency is mostly asymptomatic unless there is exposure to oxidative stress, caused by infections, or medications, followed by hemolysis [21]. There are reports of a possible association of SARS-CoV2 infection with hemolysis in adults with G6PD deficiency [22, 23]. All cases had elevated free plasma hemoglobin, elevated reticulocyte count, schistocytes on peripheral smear, but the direct antiglobulin test (DAT) was negative. About 5-10% of hemolytic anemias may have negative antibody testing on routine testing [24], due to the presence of a different type of antibody (IgM), small number of RBCs bound to IgG molecules or low-affinity autoantibodies. Additionally, during SARS-CoV-2 infection, hemolytic anemia may be DAT-negative, and traditional hemolytic markers may be unreliable [24, 25]. As seen in our cases, high plasma hemoglobin levels can be highly pathogenic through direct endothelial damage, lipid peroxidation, or activation of various inflammatory pathways.
None of the previous reports involving newborns born to SARS-CoV-2-positive mothers describe hemolytic anemia, but there are reports of autoimmune hemolytic anemia in adults with SARS-CoV-2 infection, presumably caused by the cytokine storm or through molecular mimicry [1-3, 14]. Most of the reported cases of SARS-CoV-2 infection associated with hemolytic anemia had an underlying condition predisposing to hemolysis such as spherocytosis, G6PD deficiency, or malignancy [3, 26, 27], similar to our cases. Two of the mothers have received treatment with hydroxychloroquine: for the mother of case 1 the medication was given 2 h after delivery, the mother of case 2 was started on the medication a few weeks before delivery as a treatment for lupus exacerbation, after being without this medication for many years. Hydroxychloroquine has been used in treatment of SARS-CoV-2 infections in adults, and has been linked to hemolysis in a few case reports [28]; however the use of hydroxychloroquine in pregnant women has been shown to reduce neonatal morbidity in women with systemic lupus erythematosus by significantly decreasing the rate of prematurity and IUGR without increased risk of malformations [29, 30].
Our cases highlight the need for a prospective study to identify if the incidence of hemolysis in neonates born to mothers with active or resolved SARS-CoV-2 infection during pregnancy is indeed increased. While a direct causal relationship between intrauterine exposure to SARS-CoV-2 infection or antibodies and hemolysis cannot be definitively proven, based on the clinical presentation, we speculate that infants with conditions known to cause hemolysis are at increased risk of developing a more severe form of hemolytic anemia. If intravascular hemolysis and coagulopathy are present, until further information is available, using washed blood products that are less likely to exacerbate the hemolytic process, irradiated RBC, and PCC might be a consideration.
Acknowledgments
None to declare.
Financial Disclosure
None to declare.
Conflict of Interest
The authors have no conflict of interest to disclose.
Informed Consent
Not applicable. As per our institutional IRB, no consent is required for a case report that includes up to three patients.
Author Contributions
Dr. Itriago conceptualized the manuscript, drafted the initial manuscript, and reviewed and revised the manuscript. Dr. Lingappan, Dr. Teruya and Dr. Dinu conceptualized the manuscript, reviewed and revised the manuscript. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Data Availability
The authors declare that data supporting the findings of this study are available within the article.
Abbreviations
BP: blood pressure; CPAP: continuous positive pressure; DAT: direct antiglobulin test; DIC: disseminated intravascular coagulation; DOL: day of life; G6PD: glucose-6 phosphate dehydrogenase; IgM: immunoglobulin M; IUGR: intrauterine growth restriction; LDH: lactate dehydrogenase; NEC: necrotizing enterocolitis; NICU: neonatal intensive care unit; PCC: prothrombin complex concentrate; PCR: polymerase chain reaction; RBC: red blood cell; SARS-CoV-2: severe acute respiratory syndrome coronavirus 2; TCA: T-cryptantigen
| References | ▴Top |
- Angileri F, Legare S, Marino Gammazza A, Conway de Macario E, Macario AJL, Cappello F. Is molecular mimicry the culprit in the autoimmune haemolytic anaemia affecting patients with COVID-19? Br J Haematol. 2020;190(2):e92-e93.
doi pubmed - Lazarian G, Quinquenel A, Bellal M, Siavellis J, Jacquy C, Re D, Merabet F, et al. Autoimmune haemolytic anaemia associated with COVID-19 infection. Br J Haematol. 2020;190(1):29-31.
doi pubmed - Wahlster L, Weichert-Leahey N, Trissal M, Grace RF, Sankaran VG. COVID-19 presenting with autoimmune hemolytic anemia in the setting of underlying immune dysregulation. Pediatr Blood Cancer. 2020;67(9):e28382.
doi pubmed - Nair LJ, Regukumar A, Baalamurugan KT. COVID-19-associated severe autoimmune hemolytic anemia: a rare case report. Saudi J Med Med Sci. 2021;9(3):276-279.
- Oncel MY, Akin IM, Kanburoglu MK, Tayman C, Coskun S, Narter F, Er I, et al. A multicenter study on epidemiological and clinical characteristics of 125 newborns born to women infected with COVID-19 by Turkish Neonatal Society. Eur J Pediatr. 2021;180(3):733-742.
doi pubmed - Amatya S, Corr TE, Gandhi CK, Glass KM, Kresch MJ, Mujsce DJ, Oji-Mmuo CN, et al. Management of newborns exposed to mothers with confirmed or suspected COVID-19. J Perinatol. 2020;40(7):987-996.
doi pubmed - Rad HS, Rohl J, Stylianou N, Allenby MC, Bazaz SR, Warkiani ME, Guimaraes FSF, et al. The Effects of COVID-19 on the placenta during pregnancy. Front Immunol. 2021;12:743022.
doi pubmed - Norman M, Naver L, Soderling J, Ahlberg M, Hervius Askling H, Aronsson B, Bystrom E, et al. Association of maternal SARS-CoV-2 infection in pregnancy with neonatal outcomes. JAMA. 2021;325(20):2076-2086.
doi pubmed - Garcia-Flores V, Romero R, Xu Y, Theis KR, Arenas-Hernandez M, Miller D, Peyvandipour A, et al. Maternal-fetal immune responses in pregnant women infected with SARS-CoV-2. Nat Commun. 2022;13(1):320.
doi pubmed - Vendola N, Stampini V, Amadori R, Gerbino M, Curatolo A, Surico D. Vertical transmission of antibodies in infants born from mothers with positive serology to COVID-19 pneumonia. Eur J Obstet Gynecol Reprod Biol. 2020;253:331-332.
doi pubmed - Flannery DD, Gouma S, Dhudasia MB, Mukhopadhyay S, Pfeifer MR, Woodford EC, Triebwasser JE, et al. Assessment of maternal and neonatal cord blood SARS-CoV-2 antibodies and placental transfer ratios. JAMA Pediatr. 2021;175(6):594-600.
doi pubmed - More K, Aiyer S, Goti A, Parikh M, Sheikh S, Patel G, Kallem V, et al. Multisystem inflammatory syndrome in neonates (MIS-N) associated with SARS-CoV2 infection: a case series. Eur J Pediatr. 2022.
doi pubmed - Deniz M, Tezer H. Vertical transmission of SARS CoV-2: a systematic review. J Matern Fetal Neonatal Med. 2020:1-8.
doi pubmed - Zhu H, Wang L, Fang C, Peng S, Zhang L, Chang G, Xia S, et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr. 2020;9(1):51-60.
doi pubmed - Liu P, Zheng J, Yang P, Wang X, Wei C, Zhang S, Feng S, et al. The immunologic status of newborns born to SARS-CoV-2-infected mothers in Wuhan, China. J Allergy Clin Immunol. 2020;146(1):101-109.e101.
doi pubmed - Samuels N, van de Graaf RA, de Jonge RCJ, Reiss IKM, Vermeulen MJ. Risk factors for necrotizing enterocolitis in neonates: a systematic review of prognostic studies. BMC Pediatr. 2017;17(1):105.
doi pubmed - Stout G, Lambert DK, Baer VL, Gordon PV, Henry E, Wiedmeier SE, Stoddard RA, et al. Necrotizing enterocolitis during the first week of life: a multicentered case-control and cohort comparison study. J Perinatol. 2008;28(8):556-560.
doi pubmed - Klein RL, Novak RW, Novak PE. T-cryptantigen exposure in neonatal necrotizing enterocolitis. J Pediatr Surg. 1986;21(12):1155-1158.
doi - Boralessa H, Modi N, Cockburn H, Malde R, Edwards M, Roberts I, Letsky E. RBC T activation and hemolysis in a neonatal intensive care population: implications for transfusion practice. Transfusion. 2002;42(11):1428-1434.
doi pubmed - Christensen RD, Yaish HM, Lemons RS. Neonatal hemolytic jaundice: morphologic features of erythrocytes that will help you diagnose the underlying condition. Neonatology. 2014;105(4):243-249.
doi pubmed - Nkhoma ET, Poole C, Vannappagari V, Hall SA, Beutler E. The global prevalence of glucose-6-phosphate dehydrogenase deficiency: a systematic review and meta-analysis. Blood Cells Mol Dis. 2009;42(3):267-278.
doi pubmed - Kuipers MT, van Zwieten R, Heijmans J, Rutten CE, de Heer K, Kater AP, Nur E. Glucose-6-phosphate dehydrogenase deficiency-associated hemolysis and methemoglobinemia in a COVID-19 patient treated with chloroquine. Am J Hematol. 2020;95(8):E194-E196.
doi pubmed - Maillart E, Leemans S, Van Noten H, Vandergraesen T, Mahadeb B, Salaouatchi MT, De Bels D, et al. A case report of serious haemolysis in a glucose-6-phosphate dehydrogenase-deficient COVID-19 patient receiving hydroxychloroquine. Infect Dis (Lond). 2020;52(9):659-661.
doi pubmed - Lancman G, Marcellino BK, Thibaud S, Troy K. Coombs-negative hemolytic anemia and elevated plasma hemoglobin levels in COVID-19. Ann Hematol. 2021;100(3):833-835.
doi pubmed - Garratty G. Immune hemolytic anemia associated with negative routine serology. Semin Hematol. 2005;42(3):156-164.
doi pubmed - Hindilerden F, Yonal-Hindilerden I, Akar E, Yesilbag Z, Kart-Yasar K. Severe Autoimmune Hemolytic Anemia in COVID-19 Infection, Safely Treated with Steroids. Mediterr J Hematol Infect Dis. 2020;12(1):e2020053.
doi pubmed - Severance TS, Rahim MQ, French J, 2nd, Baker RM, Shriner A, Khaitan A, Overholt KM. COVID-19 and hereditary spherocytosis: A recipe for hemolysis. Pediatr Blood Cancer. 2021;68(1):e28548.
doi pubmed - Beauverd Y, Adam Y, Assouline B, Samii K. COVID-19 infection and treatment with hydroxychloroquine cause severe haemolysis crisis in a patient with glucose-6-phosphate dehydrogenase deficiency. Eur J Haematol. 2020;105(3):357-359.
doi pubmed - Leroux M, Desveaux C, Parcevaux M, Julliac B, Gouyon JB, Dallay D, Pellegrin JL, et al. Impact of hydroxychloroquine on preterm delivery and intrauterine growth restriction in pregnant women with systemic lupus erythematosus: a descriptive cohort study. Lupus. 2015;24(13):1384-1391.
doi pubmed - Berard A, Sheehy O, Zhao JP, Vinet E, Quach C, Bernatsky S. Chloroquine and hydroxychloroquine use during pregnancy and the risk of adverse pregnancy outcomes using real-world evidence. Front Pharmacol. 2021;12:722511.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
International Journal of Clinical Pediatrics is published by Elmer Press Inc.
